|
|
|

|
Suppliers for
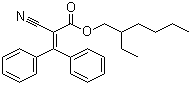
3-Diphenyl-2-cyanoacrylic acid 2-ethylhexyl ester
|
Properties | | CAS |
6197-30-4 | | Formula |
C24H27NO2 | | EINECS |
228-250-8 |
|
|
2 Registered suppliers
Product Name: Octocrilene
Synonyms: OCTYL 2-CYANO-3,3-DIPHENYLACRYLATE;
octocrilene;OCTOCRYLENE;PARSOL 340;
2-cyano-3,3-diphenyl-2-propenoicaci2-ethylhexylester;
2-ethylhexylalpha-cyano-beta-phenylcinnamate;
2-Propenoicacid,2-cyano-3,3-diphenyl-,2-ethylhexylester;
2-cyano-3,3-diphenyl-2-propanoic acid 2-ethylhexyl ester
Product Categories: cosmetic raw material;
UV-Absorber;Stabilizers;Polymer Additives;
Polymer Science
Octocrilene Chemical Properties
mp −10 °C(lit.)
bp 218 °C1.5 mm Hg(lit.)
density 1.051 g/mL at 25 °C(lit.)
refractive index n20/D 1.567(lit.)
Fp > 230 °F
General Description Yellow viscous liquid. Insoluble in water.
Air & Water Reactions Insoluble in water.
Reactivity Profile Octocrilene is an ester derivative. Esters react with acids to liberate heat along with alcohols and acids. Strong oxidizing acids may cause a vigorous reaction that is sufficiently exothermic to ignite the reaction products. Heat is also generated by the interaction of esters with caustic solutions. Flammable hydrogen is generated by mixing esters with alkali metals and hydrides.
Product Name: Octocrilene
Synonyms: OCTYL 2-CYANO-3,3-DIPHENYLACRYLATE;
octocrilene;OCTOCRYLENE;PARSOL 340;
2-cyano-3,3-diphenyl-2-propenoicaci2-ethylhexylester;
2-ethylhexylalpha-cyano-beta-phenylcinnamate;
2-Propenoicacid,2-cyano-3,3-diphenyl-,2-ethylhexylester;
2-cyano-3,3-diphenyl-2-propanoic acid 2-ethylhexyl ester
Product Categories: cosmetic raw material;
UV-Absorber;Stabilizers;Polymer Additives;
Polymer Science
Octocrilene Chemical Properties
mp −10 °C(lit.)
bp 218 °C1.5 mm Hg(lit.)
density 1.051 g/mL at 25 °C(lit.)
refractive index n20/D 1.567(lit.)
Fp > 230 °F
General Description Yellow viscous liquid. Insoluble in water.
Air & Water Reactions Insoluble in water.
Reactivity Profile Octocrilene is an ester derivative. Esters react with acids to liberate heat along with alcohols and acids. Strong oxidizing acids may cause a vigorous reaction that is sufficiently exothermic to ignite the reaction products. Heat is also generated by the interaction of esters with caustic solutions. Flammable hydrogen is generated by mixing esters with alkali metals and hydrides.
|
|
|
Privileged suppliers
Last update 2024-04-30
|


 details
details
