|
Suppliers for CAS
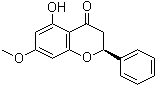
480-37-5
|
Properties | | CAS |
480-37-5 | | Formula |
C16H14O4 | | EINECS |
207-548-1 |
|
|
6 Registered suppliers
Dayang Chem (Hangzhou) Co.,Ltd.
P.R.China
Leap Chem Co., Ltd
P.R.China
Molecular Formula: C16H14O4
Molecular Weight: 270.28
Hazard Symbols: Xi
WGKGermany: 3
BOC Sciences
USA
Description : Pinostrobin, which can be extracted from the herbs of Cajanus cajan, may be responsible for the anti-hemorrhagic and analgesic activity (peripheral analgesic activity) of Renealmia alpinia. It leads to depolarisation of the cell potential of endothelial cells. Pinostrobin was able to exert a neuroprotective effect against Aβ(25-35)-induced neurotoxicity in PC12 cells, at least in part, via inhibiting oxidative damage and calcium overload, as well as suppressing the mitochondrial pathway of cellular apoptosis. Besides, Pinostrobin (50mg/kg/dose) possesses definite therapeutical effect in the development of lesion score. - Molecular Weight :270.3
- Boiling Point :494.9±45.0°C (Predicted)
- Melting Point :100°C
- Purity :> 98%
Molecular Formula : C16H14O4 Canonical SMILES : COC1=CC(=C2C(=O)CC(OC2=C1)C3=CC=CC=C3)O InChI : InChI=1S/C16H14O4/c1-19-11-7-12(17)16-13(18)9-14(20-15(16)8-11)10-5-3-2-4-6-10/h2-8,14,17H,9H2,1H3
InChIKey :
ORJDDOBAOGKRJV-UHFFFAOYSA-N
Appearance :
Powder
Application :
Anti-hemorrhagic/analgesic
Storage : 2-8°C
Synonyms :
(S)-2,3-dihydro-5-hydroxy-7-methoxy-2-phenyl-4H-1-benzopyran-4-one; (S)-2,3-dihydro-5-hydroxy-7-methoxy-2-phenyl-4-benzopyrone; 5-Hydroxy-7-methoxy-2-phenyl-chroman-4-one; 5-Hydroxy-7-methoxyflavanone; (2S)-2,3-Dihydro-5-hydroxy-7-methoxy-2-phenyl-4H-1-benzopyran-4-one; 2,3-Dihydro-5-hydroxy-7-methoxy-2-phenyl-4H-1-benzopyran-4-one; 2-Phenyl-5-hydroxy-7-methoxy-2,3-dihydro-4H-1-benzopyran-4-one
More details are to be found here
Chemos GmbH & Co. KG
Germany
Description : Pinostrobin, which can be extracted from the herbs of Cajanus cajan, may be responsible for the anti-hemorrhagic and analgesic activity (peripheral analgesic activity) of Renealmia alpinia. It leads to depolarisation of the cell potential of endothelial cells. Pinostrobin was able to exert a neuroprotective effect against Aβ(25-35)-induced neurotoxicity in PC12 cells, at least in part, via inhibiting oxidative damage and calcium overload, as well as suppressing the mitochondrial pathway of cellular apoptosis. Besides, Pinostrobin (50mg/kg/dose) possesses definite therapeutical effect in the development of lesion score. - Molecular Weight :270.3
- Boiling Point :494.9±45.0°C (Predicted)
- Melting Point :100°C
- Purity :> 98%
Molecular Formula : C16H14O4 Canonical SMILES : COC1=CC(=C2C(=O)CC(OC2=C1)C3=CC=CC=C3)O InChI : InChI=1S/C16H14O4/c1-19-11-7-12(17)16-13(18)9-14(20-15(16)8-11)10-5-3-2-4-6-10/h2-8,14,17H,9H2,1H3
InChIKey :
ORJDDOBAOGKRJV-UHFFFAOYSA-N
Appearance :
Powder
Application :
Anti-hemorrhagic/analgesic
Storage : 2-8°C
Synonyms :
(S)-2,3-dihydro-5-hydroxy-7-methoxy-2-phenyl-4H-1-benzopyran-4-one; (S)-2,3-dihydro-5-hydroxy-7-methoxy-2-phenyl-4-benzopyrone; 5-Hydroxy-7-methoxy-2-phenyl-chroman-4-one; 5-Hydroxy-7-methoxyflavanone; (2S)-2,3-Dihydro-5-hydroxy-7-methoxy-2-phenyl-4H-1-benzopyran-4-one; 2,3-Dihydro-5-hydroxy-7-methoxy-2-phenyl-4H-1-benzopyran-4-one; 2-Phenyl-5-hydroxy-7-methoxy-2,3-dihydro-4H-1-benzopyran-4-one
More details are to be found here
BuGuCh & Partners
Germany
Description : Pinostrobin, which can be extracted from the herbs of Cajanus cajan, may be responsible for the anti-hemorrhagic and analgesic activity (peripheral analgesic activity) of Renealmia alpinia. It leads to depolarisation of the cell potential of endothelial cells. Pinostrobin was able to exert a neuroprotective effect against Aβ(25-35)-induced neurotoxicity in PC12 cells, at least in part, via inhibiting oxidative damage and calcium overload, as well as suppressing the mitochondrial pathway of cellular apoptosis. Besides, Pinostrobin (50mg/kg/dose) possesses definite therapeutical effect in the development of lesion score. - Molecular Weight :270.3
- Boiling Point :494.9±45.0°C (Predicted)
- Melting Point :100°C
- Purity :> 98%
Molecular Formula : C16H14O4 Canonical SMILES : COC1=CC(=C2C(=O)CC(OC2=C1)C3=CC=CC=C3)O InChI : InChI=1S/C16H14O4/c1-19-11-7-12(17)16-13(18)9-14(20-15(16)8-11)10-5-3-2-4-6-10/h2-8,14,17H,9H2,1H3
InChIKey :
ORJDDOBAOGKRJV-UHFFFAOYSA-N
Appearance :
Powder
Application :
Anti-hemorrhagic/analgesic
Storage : 2-8°C
Synonyms :
(S)-2,3-dihydro-5-hydroxy-7-methoxy-2-phenyl-4H-1-benzopyran-4-one; (S)-2,3-dihydro-5-hydroxy-7-methoxy-2-phenyl-4-benzopyrone; 5-Hydroxy-7-methoxy-2-phenyl-chroman-4-one; 5-Hydroxy-7-methoxyflavanone; (2S)-2,3-Dihydro-5-hydroxy-7-methoxy-2-phenyl-4H-1-benzopyran-4-one; 2,3-Dihydro-5-hydroxy-7-methoxy-2-phenyl-4H-1-benzopyran-4-one; 2-Phenyl-5-hydroxy-7-methoxy-2,3-dihydro-4H-1-benzopyran-4-one
More details are to be found here
Santa Cruz Biotechnology, Inc.
USA
Description : Pinostrobin, which can be extracted from the herbs of Cajanus cajan, may be responsible for the anti-hemorrhagic and analgesic activity (peripheral analgesic activity) of Renealmia alpinia. It leads to depolarisation of the cell potential of endothelial cells. Pinostrobin was able to exert a neuroprotective effect against Aβ(25-35)-induced neurotoxicity in PC12 cells, at least in part, via inhibiting oxidative damage and calcium overload, as well as suppressing the mitochondrial pathway of cellular apoptosis. Besides, Pinostrobin (50mg/kg/dose) possesses definite therapeutical effect in the development of lesion score. - Molecular Weight :270.3
- Boiling Point :494.9±45.0°C (Predicted)
- Melting Point :100°C
- Purity :> 98%
Molecular Formula : C16H14O4 Canonical SMILES : COC1=CC(=C2C(=O)CC(OC2=C1)C3=CC=CC=C3)O InChI : InChI=1S/C16H14O4/c1-19-11-7-12(17)16-13(18)9-14(20-15(16)8-11)10-5-3-2-4-6-10/h2-8,14,17H,9H2,1H3
InChIKey :
ORJDDOBAOGKRJV-UHFFFAOYSA-N
Appearance :
Powder
Application :
Anti-hemorrhagic/analgesic
Storage : 2-8°C
Synonyms :
(S)-2,3-dihydro-5-hydroxy-7-methoxy-2-phenyl-4H-1-benzopyran-4-one; (S)-2,3-dihydro-5-hydroxy-7-methoxy-2-phenyl-4-benzopyrone; 5-Hydroxy-7-methoxy-2-phenyl-chroman-4-one; 5-Hydroxy-7-methoxyflavanone; (2S)-2,3-Dihydro-5-hydroxy-7-methoxy-2-phenyl-4H-1-benzopyran-4-one; 2,3-Dihydro-5-hydroxy-7-methoxy-2-phenyl-4H-1-benzopyran-4-one; 2-Phenyl-5-hydroxy-7-methoxy-2,3-dihydro-4H-1-benzopyran-4-one
More details are to be found here
|


 details
details details
details_Co_Ltd_1_575.jpg)



