|
|
|

|
Suppliers for
1,4-Butane dicarboxylic acid
|
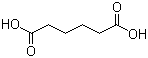
Properties | | CAS |
124-04-9 | | Formula |
C6H10O4 |
|
|
1 Registered suppliers
Product Name: Adipic acid
Synonyms:
RARECHEM AL BO 0180;AKOS BBS-00004308;ADIPIC ACID;adipinic acid;1,6-HEXANEDIOIC ACID;1,4-BUTANEDICARBOXYLIC ACID;BUTANE-1,4-DICARBOXYLIC ACID;DICARBOXYLIC ACID C6
CAS:124-04-9
MF:C6H10O4
MW:146.14
EINECS:204-673-3
Melting point:151-154 °C(lit.)
Boiling point: 265 °C100 mm Hg(lit.)
Density: 1,36 g/cm3
storage temp. : in R.T.
Stability: Stable. Substances to be avoided include ammonia, strong oxidizing agents.
Package: 25kg/BAG
Purity: 99%
Chemical properties:
White crystals, with scorched bones. Slightly soluble in water, soluble in alcohol, ether and most organic solvents.
Usage:
Mainly used for nylon 66 and engineering plastics raw materials, but also for the production of various ester products, but also for the polyurethane elastomer raw materials
For the synthesis of polymer raw materials, but also for the system of plasticizers and lubricants
Organic synthesis, flux, resin, plastic.
Brief manufacture process:
I Cyclohexane One-step oxidation
Cyclohexane as raw material, acetic acid as solvent, cobalt and bromide as a catalyst, in 2MP. And 90 ° C for 10 ~ 13h. Yield 75%.
2 Cyclohexane step oxidation method
(1) Preparation of KA oil can be 1.0 ~ 2.5MPa and 145 ~ 180 ℃ with air direct oxidation, the yield of 70% to 75%. Can also be used as a catalyst, 1.0 ~ 2.0MPa and 165 ℃ under air oxidation, the yield of 90%, alcohol to ketone ratio of 10: 1; reactants with hot water treatment, ester hydrolysis, stratification, water Layer to recover boric acid, dehydrated into boric acid recycling; organic layer saponified with organic sodium caustic soda, and remove the acid, distillation cyclohexane recovery after alcohol ketone mixture.
(2) Oxidation of KA oil KA oil was oxidized in an excess of 50% to 60% nitric acid in a two-stage series reactor at 60-80C and 0.1-0.4 MPa. The catalyst is copper-vanadium (copper 0.1% to 0.5%, vanadium 0.1% to 0.2%) and the yield is 92% to 96% of the theoretical value. After the reactant is distilled out of nitric acid, the high-purity adipic acid can be obtained by twice crystallization purification.
|
|
|
www.BuyersGuideChem.de
is visited by more than
75,000 readers
per month
We are online since 1996
Last update 2024-04-26
|


 details
details


