|
|
|

|
Suppliers for
Pseudoginsenoside - F11
|
Properties | | CAS |
69884-00-0 | | Formula |
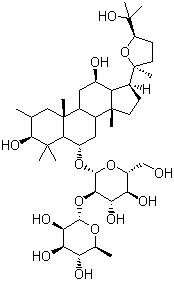
C42H72O14 |
|
|
7 Registered suppliers
Molecular Formula: C42H72O14 Molecular Weight: 801,01
More details are to be found here
Description : Pseudoginsenoside- F11 is extracted from the roots of Panax ginseng C. A. Mey. It antagonized the memory dysfunction induced by scopolamine. It has been shown to antagonize the behavioral actions of morphine. It may block the development of morphine-induced behavioral sensitization via its effect, at least partially, on the glutamatergic system in the medial prefrontal cortex (mPFC). - Molecular Weight :801.01
- Melting Point :> 196 °C
- Purity :> 98%
Molecular Formula : C42H72O14 Canonical SMILES : CC1C(C(C(C(O1)OC2C(C(C(OC2OC3CC4(C(CC(C5C4(CCC5C6(CCC(O6)C(C)(C)O)C)C)O)C7(C3C(C(CC7)O)(C)C)C)C)CO)O)O)O)O)O
InChI :
1S/C42H72O14/c1-19-28(46)30(48)32(50)35(52-19)55-33-31(49)29(47)23(18-43)54-36(33)53-22-17-41(8)24(39(6)13-11-25(45)37(2,3)34(22)39)16-21(44)27-20(10-14-40(27,41)7)42(9)15-12-26(56-42)38(4,5)51/h19-36,43-51H,10-18H2,1-9H3/t19-,20-,21+,22-,23+,24+,25-,26+,
InChIKey :
JBGYSAVRIDZNKA-SQBONPAESA-N
Solubility :
Soluble in DMSO
Appearance :
White powder
Application :
An oxotillol-type ginsenoside which displayes neuroprotective activity.
Storage : Store at -20 °C
Synonyms :
(3b,6a,12b,24R)-20,24-Epoxy-3,12,25-trihydroxydammaran-6-yl 2-O-(6-deoxy-alpha-L-mannopyranosyl)-beta-D-glucopyranoside; (20S,24R)-6α-(2-O-α-L-Rhamnopyranosyl-β-D-glucopyranosyloxy)-20,24-epoxy-5α-dammarane-3β,12β,25-triol; (24R)-Pseudoginsenoside F11; [(24R)-20,24-Epoxy-3β,12β,25-trihydroxy-5α-dammaran-6α-yl]2-O-(6-deoxy-α-L-mannopyranosyl)-β-D-glucopyranoside; 17β-[[(2S)-2β-Methyl-5α-(1-hydroxy-1-methylethyl)tetrahydrofuran]-2-yl]-3β,12β-dihydroxy-4,4,8β,10β,14α-pentamethyl-5α-gonane-6α-yl 2-O-α-L-rhamnopyranosyl-β-D-glucopyranoside; Ginsenoside A1
More details are to be found here
Description : Pseudoginsenoside- F11 is extracted from the roots of Panax ginseng C. A. Mey. It antagonized the memory dysfunction induced by scopolamine. It has been shown to antagonize the behavioral actions of morphine. It may block the development of morphine-induced behavioral sensitization via its effect, at least partially, on the glutamatergic system in the medial prefrontal cortex (mPFC). - Molecular Weight :801.01
- Melting Point :> 196 °C
- Purity :> 98%
Molecular Formula : C42H72O14 Canonical SMILES : CC1C(C(C(C(O1)OC2C(C(C(OC2OC3CC4(C(CC(C5C4(CCC5C6(CCC(O6)C(C)(C)O)C)C)O)C7(C3C(C(CC7)O)(C)C)C)C)CO)O)O)O)O)O
InChI :
1S/C42H72O14/c1-19-28(46)30(48)32(50)35(52-19)55-33-31(49)29(47)23(18-43)54-36(33)53-22-17-41(8)24(39(6)13-11-25(45)37(2,3)34(22)39)16-21(44)27-20(10-14-40(27,41)7)42(9)15-12-26(56-42)38(4,5)51/h19-36,43-51H,10-18H2,1-9H3/t19-,20-,21+,22-,23+,24+,25-,26+,
InChIKey :
JBGYSAVRIDZNKA-SQBONPAESA-N
Solubility :
Soluble in DMSO
Appearance :
White powder
Application :
An oxotillol-type ginsenoside which displayes neuroprotective activity.
Storage : Store at -20 °C
Synonyms :
(3b,6a,12b,24R)-20,24-Epoxy-3,12,25-trihydroxydammaran-6-yl 2-O-(6-deoxy-alpha-L-mannopyranosyl)-beta-D-glucopyranoside; (20S,24R)-6α-(2-O-α-L-Rhamnopyranosyl-β-D-glucopyranosyloxy)-20,24-epoxy-5α-dammarane-3β,12β,25-triol; (24R)-Pseudoginsenoside F11; [(24R)-20,24-Epoxy-3β,12β,25-trihydroxy-5α-dammaran-6α-yl]2-O-(6-deoxy-α-L-mannopyranosyl)-β-D-glucopyranoside; 17β-[[(2S)-2β-Methyl-5α-(1-hydroxy-1-methylethyl)tetrahydrofuran]-2-yl]-3β,12β-dihydroxy-4,4,8β,10β,14α-pentamethyl-5α-gonane-6α-yl 2-O-α-L-rhamnopyranosyl-β-D-glucopyranoside; Ginsenoside A1
More details are to be found here
Description : Pseudoginsenoside- F11 is extracted from the roots of Panax ginseng C. A. Mey. It antagonized the memory dysfunction induced by scopolamine. It has been shown to antagonize the behavioral actions of morphine. It may block the development of morphine-induced behavioral sensitization via its effect, at least partially, on the glutamatergic system in the medial prefrontal cortex (mPFC). - Molecular Weight :801.01
- Melting Point :> 196 °C
- Purity :> 98%
Molecular Formula : C42H72O14 Canonical SMILES : CC1C(C(C(C(O1)OC2C(C(C(OC2OC3CC4(C(CC(C5C4(CCC5C6(CCC(O6)C(C)(C)O)C)C)O)C7(C3C(C(CC7)O)(C)C)C)C)CO)O)O)O)O)O
InChI :
1S/C42H72O14/c1-19-28(46)30(48)32(50)35(52-19)55-33-31(49)29(47)23(18-43)54-36(33)53-22-17-41(8)24(39(6)13-11-25(45)37(2,3)34(22)39)16-21(44)27-20(10-14-40(27,41)7)42(9)15-12-26(56-42)38(4,5)51/h19-36,43-51H,10-18H2,1-9H3/t19-,20-,21+,22-,23+,24+,25-,26+,
InChIKey :
JBGYSAVRIDZNKA-SQBONPAESA-N
Solubility :
Soluble in DMSO
Appearance :
White powder
Application :
An oxotillol-type ginsenoside which displayes neuroprotective activity.
Storage : Store at -20 °C
Synonyms :
(3b,6a,12b,24R)-20,24-Epoxy-3,12,25-trihydroxydammaran-6-yl 2-O-(6-deoxy-alpha-L-mannopyranosyl)-beta-D-glucopyranoside; (20S,24R)-6α-(2-O-α-L-Rhamnopyranosyl-β-D-glucopyranosyloxy)-20,24-epoxy-5α-dammarane-3β,12β,25-triol; (24R)-Pseudoginsenoside F11; [(24R)-20,24-Epoxy-3β,12β,25-trihydroxy-5α-dammaran-6α-yl]2-O-(6-deoxy-α-L-mannopyranosyl)-β-D-glucopyranoside; 17β-[[(2S)-2β-Methyl-5α-(1-hydroxy-1-methylethyl)tetrahydrofuran]-2-yl]-3β,12β-dihydroxy-4,4,8β,10β,14α-pentamethyl-5α-gonane-6α-yl 2-O-α-L-rhamnopyranosyl-β-D-glucopyranoside; Ginsenoside A1
More details are to be found here
Description : Pseudoginsenoside- F11 is extracted from the roots of Panax ginseng C. A. Mey. It antagonized the memory dysfunction induced by scopolamine. It has been shown to antagonize the behavioral actions of morphine. It may block the development of morphine-induced behavioral sensitization via its effect, at least partially, on the glutamatergic system in the medial prefrontal cortex (mPFC). - Molecular Weight :801.01
- Melting Point :> 196 °C
- Purity :> 98%
Molecular Formula : C42H72O14 Canonical SMILES : CC1C(C(C(C(O1)OC2C(C(C(OC2OC3CC4(C(CC(C5C4(CCC5C6(CCC(O6)C(C)(C)O)C)C)O)C7(C3C(C(CC7)O)(C)C)C)C)CO)O)O)O)O)O
InChI :
1S/C42H72O14/c1-19-28(46)30(48)32(50)35(52-19)55-33-31(49)29(47)23(18-43)54-36(33)53-22-17-41(8)24(39(6)13-11-25(45)37(2,3)34(22)39)16-21(44)27-20(10-14-40(27,41)7)42(9)15-12-26(56-42)38(4,5)51/h19-36,43-51H,10-18H2,1-9H3/t19-,20-,21+,22-,23+,24+,25-,26+,
InChIKey :
JBGYSAVRIDZNKA-SQBONPAESA-N
Solubility :
Soluble in DMSO
Appearance :
White powder
Application :
An oxotillol-type ginsenoside which displayes neuroprotective activity.
Storage : Store at -20 °C
Synonyms :
(3b,6a,12b,24R)-20,24-Epoxy-3,12,25-trihydroxydammaran-6-yl 2-O-(6-deoxy-alpha-L-mannopyranosyl)-beta-D-glucopyranoside; (20S,24R)-6α-(2-O-α-L-Rhamnopyranosyl-β-D-glucopyranosyloxy)-20,24-epoxy-5α-dammarane-3β,12β,25-triol; (24R)-Pseudoginsenoside F11; [(24R)-20,24-Epoxy-3β,12β,25-trihydroxy-5α-dammaran-6α-yl]2-O-(6-deoxy-α-L-mannopyranosyl)-β-D-glucopyranoside; 17β-[[(2S)-2β-Methyl-5α-(1-hydroxy-1-methylethyl)tetrahydrofuran]-2-yl]-3β,12β-dihydroxy-4,4,8β,10β,14α-pentamethyl-5α-gonane-6α-yl 2-O-α-L-rhamnopyranosyl-β-D-glucopyranoside; Ginsenoside A1
More details are to be found here
Description : Pseudoginsenoside- F11 is extracted from the roots of Panax ginseng C. A. Mey. It antagonized the memory dysfunction induced by scopolamine. It has been shown to antagonize the behavioral actions of morphine. It may block the development of morphine-induced behavioral sensitization via its effect, at least partially, on the glutamatergic system in the medial prefrontal cortex (mPFC). - Molecular Weight :801.01
- Melting Point :> 196 °C
- Purity :> 98%
Molecular Formula : C42H72O14 Canonical SMILES : CC1C(C(C(C(O1)OC2C(C(C(OC2OC3CC4(C(CC(C5C4(CCC5C6(CCC(O6)C(C)(C)O)C)C)O)C7(C3C(C(CC7)O)(C)C)C)C)CO)O)O)O)O)O
InChI :
1S/C42H72O14/c1-19-28(46)30(48)32(50)35(52-19)55-33-31(49)29(47)23(18-43)54-36(33)53-22-17-41(8)24(39(6)13-11-25(45)37(2,3)34(22)39)16-21(44)27-20(10-14-40(27,41)7)42(9)15-12-26(56-42)38(4,5)51/h19-36,43-51H,10-18H2,1-9H3/t19-,20-,21+,22-,23+,24+,25-,26+,
InChIKey :
JBGYSAVRIDZNKA-SQBONPAESA-N
Solubility :
Soluble in DMSO
Appearance :
White powder
Application :
An oxotillol-type ginsenoside which displayes neuroprotective activity.
Storage : Store at -20 °C
Synonyms :
(3b,6a,12b,24R)-20,24-Epoxy-3,12,25-trihydroxydammaran-6-yl 2-O-(6-deoxy-alpha-L-mannopyranosyl)-beta-D-glucopyranoside; (20S,24R)-6α-(2-O-α-L-Rhamnopyranosyl-β-D-glucopyranosyloxy)-20,24-epoxy-5α-dammarane-3β,12β,25-triol; (24R)-Pseudoginsenoside F11; [(24R)-20,24-Epoxy-3β,12β,25-trihydroxy-5α-dammaran-6α-yl]2-O-(6-deoxy-α-L-mannopyranosyl)-β-D-glucopyranoside; 17β-[[(2S)-2β-Methyl-5α-(1-hydroxy-1-methylethyl)tetrahydrofuran]-2-yl]-3β,12β-dihydroxy-4,4,8β,10β,14α-pentamethyl-5α-gonane-6α-yl 2-O-α-L-rhamnopyranosyl-β-D-glucopyranoside; Ginsenoside A1
More details are to be found here
|
|
|
Privileged suppliers
Last update 2024-06-21
|


 details
details details
details_Co_Ltd_1_575.jpg)




