|
|
|

|
Suppliers for
Avibactam sodium salt
|
Properties | | CAS |
1192491-61-4 |
|
|
9 Registered suppliers
More details are to be found here
More details are to be found here
More details are to be found here
More details are to be found here
More details are to be found here
More details are to be found here
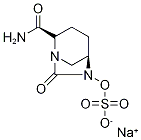
Description : Avibactam Sodium Salt, a novel non-ᵦ-lactam ᵦ-lactamase inhibitor, binds covalently to Ambler class A ᵦ-lactamases, including KPCs, Ambler class C and some class D b-lactamases. In combination with ceftazidime, Avibactam is a new drug application for avibactam , and was approved by the FDA on February 25, 2015, to treat complicated urinary tract and complicated intra-abdominal Infections caused by antibiotic resistant-pathogens. Avibactam has limited intrinsic antimicrobial activity but restores the in vitro activity of b-lactams including ceftazidime, ceftaroline, the active metabolite of ceftaroline fosamil, and aztreonam against ESBL-producing pathogens. - Molecular Weight :287.23
- Melting Point :> 208°C (dec.)
Molecular Formula : C7H10N3NaO6S Canonical SMILES : O=C1N(OS(=O)(O[Na])=O)[C@H]2CN1[C@H](C(N)=O)CC2 InChI : 1S/C7H11N3O6S.Na/c8-6(11)5-2-1-4-3-9(5)7(12)10(4)16-17(13,14)15;/h4-5H,1-3H2,(H2,8,11)(H,13,14,15);/q;+1/p-1/t4-,5+;/m1./s1
InChIKey :
RTCIKUMODPANKX-JBUOLDKXSA-M
Solubility :
Soluble in DMSO (slightly), methanol (slightly, sonicated), water (slightly)
Appearance :
Solid powder
Storage : Store in a cool and dry place and at 0 - 4℃ for short term (days to weeks) or -42℃ for long term (months to years).
Synonyms :
sodium (2S,5R)-2-carbamoyl-7-oxo-1,6-diazabicyclo[321]octan-6-yl sulfate; NXL104; NXL-104; NXL 104; Avibactam; Avibactam sodium
More details are to be found here
Description : Avibactam Sodium Salt, a novel non-ᵦ-lactam ᵦ-lactamase inhibitor, binds covalently to Ambler class A ᵦ-lactamases, including KPCs, Ambler class C and some class D b-lactamases. In combination with ceftazidime, Avibactam is a new drug application for avibactam , and was approved by the FDA on February 25, 2015, to treat complicated urinary tract and complicated intra-abdominal Infections caused by antibiotic resistant-pathogens. Avibactam has limited intrinsic antimicrobial activity but restores the in vitro activity of b-lactams including ceftazidime, ceftaroline, the active metabolite of ceftaroline fosamil, and aztreonam against ESBL-producing pathogens. - Molecular Weight :287.23
- Melting Point :> 208°C (dec.)
Molecular Formula : C7H10N3NaO6S Canonical SMILES : O=C1N(OS(=O)(O[Na])=O)[C@H]2CN1[C@H](C(N)=O)CC2 InChI : 1S/C7H11N3O6S.Na/c8-6(11)5-2-1-4-3-9(5)7(12)10(4)16-17(13,14)15;/h4-5H,1-3H2,(H2,8,11)(H,13,14,15);/q;+1/p-1/t4-,5+;/m1./s1
InChIKey :
RTCIKUMODPANKX-JBUOLDKXSA-M
Solubility :
Soluble in DMSO (slightly), methanol (slightly, sonicated), water (slightly)
Appearance :
Solid powder
Storage : Store in a cool and dry place and at 0 - 4℃ for short term (days to weeks) or -42℃ for long term (months to years).
Synonyms :
sodium (2S,5R)-2-carbamoyl-7-oxo-1,6-diazabicyclo[321]octan-6-yl sulfate; NXL104; NXL-104; NXL 104; Avibactam; Avibactam sodium
More details are to be found here
Purity : 98% Smile code : O=S(ON1[C@]2([H])CC[C@@H](C(N)=O)[N@@](C2)C1=O)([O-])=O.[Na+]
MDL Number : MFCD28900719
MolFormula : C7H10N3NaO6S
MolWeight : 287.2256
Available in stock : 143.156
More details are to be found here
|
|
|
Privileged suppliers
Last update 2024-05-28
|


 details
details details
details details
details







