|
|
|

|
Proveedores de
Sumatriptan succinate
|
Identificación | | CAS |
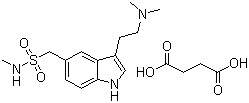
103628-48-4 | | Formula |
C14H21N3O2S.C4H6O4 |
|
|
16 Proveedores registrados
| Character |
| | Appearance | White or almost white powder.
| | Solubility | Freely soluble in water, sparingly soluble in methanol, practically insoluble in methylene chloride.
| | PH | 4.5~ 5.3
| | Absorbance | Maximum 0.10 percent
| | Related substances |
| | Impurity A | not more than 0.6 percent
| | Impurity H | not more than 0.3 percent
| | Impurity B,C or D | each impurity not more than 0.5 percent
| | Impurity E or any other impurity | each impurities not more than 0.1 percent
| | Total impurities | not more than 0.6 percent
| | Water | not more than 1.0 percent
| | Sulphated ash | not more than 0.1 percent
| | Residual solvents |
| | Methanol | not more than 3000ppm
| | Acetonitrite | not more than 100ppm
| | Isopropyl Alcohol | not more than 100ppm
| | Chloroform | not more than 60ppm
| | Assay | 97.5 percent to102.0 percent (Anhydrous substance).
| | Standard | EP standard
| | Content | 97.5~102.0%
| | Capacity | 30MT/month
| | Loading port | Shanghai
| | Use | Sumatriptan Succinate is an active pharmaceutical ingredient used for migraine
| | Package | 25kg/drum or according to the customers’ demand
| | Storage | Stored in the dry and ventilated inside storeroom, prevent direct sunlight, slightly pile and put down |
Molecular Formula: C14H21N3O2S.C4H6O4 Molecular Weight: 413,49 Más detalles se encuentran aquí
|
|
|
Proveedores
privilegiados
Última actualización
2024-05-10 |