|
Suppliers for CAS
79233-15-1
|
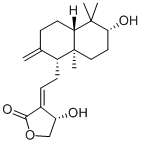
Properties | | CAS |
79233-15-1 | | Formula |
C20H30O4 |
|
|
4 Registered suppliers
Dayang Chem (Hangzhou) Co.,Ltd.
P.R.China
Leap Chem Co., Ltd
P.R.China
Molecular Formula: C20H30O4
Molecular Weight: 334.45
Molecular Formula: C20H30O4 Molecular Weight: 334,45
More details are to be found here
BOC Sciences
USA
Description : Deoxyandrographolide is a natural compound extracted from the herbs of Andrographis paniculata (Burm. f.) Nees. It could potently inhibit the growth of liver (HepG2 and SK-Hep1) and bile duct (HuCCA-1 and RMCCA-1) cancer cells. It controlled ethanol-induced hepatosteatosis by interfering with dysregulation of lipid metabolism. It was capable of preventing the development of fatty liver through AMPK-mediated regulation of lipid metabolism. It reduced the extracellular acidification rate and the intracellular alkalinization in a dose-dependent manner in concentrations between 10-100 microM. It reduced PAF-induced calcium flux in the presence of extracellular calcium. It mediated activation of adenylate cyclase-cAMP signaling leading to up-regulation of cNOS may provide a promising approach in the prevention of liver diseases during chronic alcoholism. - Molecular Weight :334.45
- Boiling Point :511.7±50.0 °C | Condition: Press: 760 Torr
- Purity :> 98%
Molecular Formula : C20H30O4 Canonical SMILES : CC1=C(C2(CCC(C(C2CC1)(C)CO)O)C)CCC3=CCOC3=O InChI : InChI=1S/C20H30O4/c1-13-4-7-16-19(2,10-8-17(22)20(16,3)12-21)15(13)6-5-14-9-11-24-18(14)23/h9,16-17,21-22H,4-8,10-12H2,1-3H3/t16-,17+,19-,20-/m0/s1
InChIKey :
DAXSYTVXDSOSIE-HNJRGHQBSA-N
Solubility :
10 mM in DMSO
Appearance :
Solid powder
Application :
Deoxyandrographolide could potently inhibit the growth of liver (HepG2 and SK-Hep1) and bile duct (HuCCA-1 and RMCCA-1) cancer cells. It controlled ethanol-induced hepatosteatosis by interfering with dysregulation of lipid metabolism. It reduced the extracellular acidification rate and the intracellular alkalinization in a dose-dependent manner in concentrations between 10-100 microM. It reduced PAF-induced calcium flux in the presence of extracellular calcium.
Storage : -20°C Freezer
Synonyms :
4-[2-[(4aS,5R,6R,8aR)-6-hydroxy-5-(hydroxymethyl)-2,5,8a-trimethyl-3,4,4a,6,7,8-hexahydronaphthalen-1-yl]ethyl]-2H-furan-5-one;2(5H)-Furanone, 3-[2-[(4aS,5R,6R,8aR)-3,4,4a,5,6,7,8,8a-octahydro-6-hydroxy-5-(hydroxymethyl)-2,5,8a-trimethyl-1-naphthalenyl]ethyl]-
More details are to be found here
Chemos GmbH & Co. KG
Germany
Description : Deoxyandrographolide is a natural compound extracted from the herbs of Andrographis paniculata (Burm. f.) Nees. It could potently inhibit the growth of liver (HepG2 and SK-Hep1) and bile duct (HuCCA-1 and RMCCA-1) cancer cells. It controlled ethanol-induced hepatosteatosis by interfering with dysregulation of lipid metabolism. It was capable of preventing the development of fatty liver through AMPK-mediated regulation of lipid metabolism. It reduced the extracellular acidification rate and the intracellular alkalinization in a dose-dependent manner in concentrations between 10-100 microM. It reduced PAF-induced calcium flux in the presence of extracellular calcium. It mediated activation of adenylate cyclase-cAMP signaling leading to up-regulation of cNOS may provide a promising approach in the prevention of liver diseases during chronic alcoholism. - Molecular Weight :334.45
- Boiling Point :511.7±50.0 °C | Condition: Press: 760 Torr
- Purity :> 98%
Molecular Formula : C20H30O4 Canonical SMILES : CC1=C(C2(CCC(C(C2CC1)(C)CO)O)C)CCC3=CCOC3=O InChI : InChI=1S/C20H30O4/c1-13-4-7-16-19(2,10-8-17(22)20(16,3)12-21)15(13)6-5-14-9-11-24-18(14)23/h9,16-17,21-22H,4-8,10-12H2,1-3H3/t16-,17+,19-,20-/m0/s1
InChIKey :
DAXSYTVXDSOSIE-HNJRGHQBSA-N
Solubility :
10 mM in DMSO
Appearance :
Solid powder
Application :
Deoxyandrographolide could potently inhibit the growth of liver (HepG2 and SK-Hep1) and bile duct (HuCCA-1 and RMCCA-1) cancer cells. It controlled ethanol-induced hepatosteatosis by interfering with dysregulation of lipid metabolism. It reduced the extracellular acidification rate and the intracellular alkalinization in a dose-dependent manner in concentrations between 10-100 microM. It reduced PAF-induced calcium flux in the presence of extracellular calcium.
Storage : -20°C Freezer
Synonyms :
4-[2-[(4aS,5R,6R,8aR)-6-hydroxy-5-(hydroxymethyl)-2,5,8a-trimethyl-3,4,4a,6,7,8-hexahydronaphthalen-1-yl]ethyl]-2H-furan-5-one;2(5H)-Furanone, 3-[2-[(4aS,5R,6R,8aR)-3,4,4a,5,6,7,8,8a-octahydro-6-hydroxy-5-(hydroxymethyl)-2,5,8a-trimethyl-1-naphthalenyl]ethyl]-
More details are to be found here
|


 details
details details
details_Co_Ltd_1_575.jpg)


