|
|
|
|
Suppliers for
Ceftriaxone sodium sterile
|
Properties | | CAS |
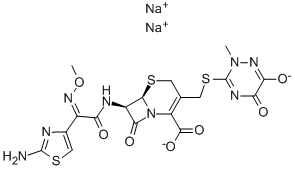
104376-79-6 | | Formula |
C18H17N8NaO7S3 |
|
|
2 Registered suppliers
| Appearance | White to pale yellow Crystalline Powder
| | Identification | IR Spectrum
| | Clarity | ≤ 1#Clarty standard solution
| | Crystallinity | Meets the requirements
| | PH | 6.0~8.0
| | Water | 8.0%~11.0%
| | Sterility | Sterile
| | Bacterial Endotoxins | NMT 0.20 USP EU/mg
| | Acetone | ≤ 0.5%
| | Viable matter | ≤ 5
| | Particulate matter | ≥ 10um NMT 6000 ≥ 25 um NMT 600
| | Assay (HPLC) | ≥ 795ug/mg (as Ceftriaxone, anhydrous) |
| Appearance | White to pale yellow Crystalline Powder
| | Identification | IR Spectrum
| | Clarity | ≤ 1#Clarty standard solution
| | Crystallinity | Meets the requirements
| | PH | 6.0~8.0
| | Water | 8.0%~11.0%
| | Sterility | Sterile
| | Bacterial Endotoxins | NMT 0.20 USP EU/mg
| | Acetone | ≤ 0.5%
| | Viable matter | ≤ 5
| | Particulate matter | ≥ 10um NMT 6000 ≥ 25 um NMT 600
| | Assay (HPLC) | ≥ 795ug/mg (as Ceftriaxone, anhydrous) |
|
|
|
Privileged suppliers
Last update 2024-04-26
|


 details
details
