|
|
|

|
Suppliers for
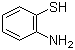
2-Aminothiophenol
|
Properties | | CAS |
137-07-5 | | Formula |
C6H7NS | | EINECS |
205-277-3 |
|
|
18 Registered suppliers
2-Aminothiophenol is an organosulfur compound with the formula C6H4(SH)(NH2). It is a colorless oily solid, although impure samples can be deeply colored. It is soluble in organic solvents and in basic water. It is a precursor to benzothiazoles, some of which are bioactive or are dyes. Isomers of aminothiophenols include 3-aminothiophenol and 4-aminothiophenol.
2-Aminothiophenol can prepared in two steps, starting with the reaction of aniline with carbon disulfide followed by hydrolysis of the resulting mercaptobenzothiazole. It can also obtained by zinc reduction of 2-nitrobenzenesulfonyl chloride.
| | Appearance & Physical State | clear yellow to amber liquid
| | Density | 1.16
| | Melting Point | 23-26ºC
| | Boiling Point | 234ºC
| | Flash Point | 79ºC
| | Refractive Index | 1.64-1.644
| | Water Solubility | INSOLUBLE
| | Stability | Stable at room temperature in closed containers under normal storage and handling conditions.
| | Storage Condition | Keep away from heat, sparks, and flame. Keep away from sources of ignition. Keep container closed when not in use. Store in a cool, dry, well-ventilated area away from incompatible substances. Corrosives area. Do not expose to air. Store under an inert atm
| | Vapor Pressure | 0.0517mmHg at 25°C |
2-Aminothiophenol is an organosulfur compound with the formula C6H4(SH)(NH2). It is a colorless oily solid, although impure samples can be deeply colored. It is soluble in organic solvents and in basic water. It is a precursor to benzothiazoles, some of which are bioactive or are dyes. Isomers of aminothiophenols include 3-aminothiophenol and 4-aminothiophenol.
2-Aminothiophenol can prepared in two steps, starting with the reaction of aniline with carbon disulfide followed by hydrolysis of the resulting mercaptobenzothiazole. It can also obtained by zinc reduction of 2-nitrobenzenesulfonyl chloride.
| | Appearance & Physical State | clear yellow to amber liquid
| | Density | 1.16
| | Melting Point | 23-26ºC
| | Boiling Point | 234ºC
| | Flash Point | 79ºC
| | Refractive Index | 1.64-1.644
| | Water Solubility | INSOLUBLE
| | Stability | Stable at room temperature in closed containers under normal storage and handling conditions.
| | Storage Condition | Keep away from heat, sparks, and flame. Keep away from sources of ignition. Keep container closed when not in use. Store in a cool, dry, well-ventilated area away from incompatible substances. Corrosives area. Do not expose to air. Store under an inert atm
| | Vapor Pressure | 0.0517mmHg at 25°C |
2-Aminothiophenol is an organosulfur compound with the formula C6H4(SH)(NH2). It is a colorless oily solid, although impure samples can be deeply colored. It is soluble in organic solvents and in basic water. It is a precursor to benzothiazoles, some of which are bioactive or are dyes. Isomers of aminothiophenols include 3-aminothiophenol and 4-aminothiophenol.
2-Aminothiophenol can prepared in two steps, starting with the reaction of aniline with carbon disulfide followed by hydrolysis of the resulting mercaptobenzothiazole. It can also obtained by zinc reduction of 2-nitrobenzenesulfonyl chloride.
| | Appearance & Physical State | clear yellow to amber liquid
| | Density | 1.16
| | Melting Point | 23-26ºC
| | Boiling Point | 234ºC
| | Flash Point | 79ºC
| | Refractive Index | 1.64-1.644
| | Water Solubility | INSOLUBLE
| | Stability | Stable at room temperature in closed containers under normal storage and handling conditions.
| | Storage Condition | Keep away from heat, sparks, and flame. Keep away from sources of ignition. Keep container closed when not in use. Store in a cool, dry, well-ventilated area away from incompatible substances. Corrosives area. Do not expose to air. Store under an inert atm
| | Vapor Pressure | 0.0517mmHg at 25°C |
2-Aminothiophenol is an organosulfur compound with the formula C6H4(SH)(NH2). It is a colorless oily solid, although impure samples can be deeply colored. It is soluble in organic solvents and in basic water. It is a precursor to benzothiazoles, some of which are bioactive or are dyes. Isomers of aminothiophenols include 3-aminothiophenol and 4-aminothiophenol.
2-Aminothiophenol can prepared in two steps, starting with the reaction of aniline with carbon disulfide followed by hydrolysis of the resulting mercaptobenzothiazole. It can also obtained by zinc reduction of 2-nitrobenzenesulfonyl chloride.
| | Appearance & Physical State | clear yellow to amber liquid
| | Density | 1.16
| | Melting Point | 23-26ºC
| | Boiling Point | 234ºC
| | Flash Point | 79ºC
| | Refractive Index | 1.64-1.644
| | Water Solubility | INSOLUBLE
| | Stability | Stable at room temperature in closed containers under normal storage and handling conditions.
| | Storage Condition | Keep away from heat, sparks, and flame. Keep away from sources of ignition. Keep container closed when not in use. Store in a cool, dry, well-ventilated area away from incompatible substances. Corrosives area. Do not expose to air. Store under an inert atm
| | Vapor Pressure | 0.0517mmHg at 25°C |
2-Aminothiophenol is an organosulfur compound with the formula C6H4(SH)(NH2). It is a colorless oily solid, although impure samples can be deeply colored. It is soluble in organic solvents and in basic water. It is a precursor to benzothiazoles, some of which are bioactive or are dyes. Isomers of aminothiophenols include 3-aminothiophenol and 4-aminothiophenol.
2-Aminothiophenol can prepared in two steps, starting with the reaction of aniline with carbon disulfide followed by hydrolysis of the resulting mercaptobenzothiazole. It can also obtained by zinc reduction of 2-nitrobenzenesulfonyl chloride.
| | Appearance & Physical State | clear yellow to amber liquid
| | Density | 1.16
| | Melting Point | 23-26ºC
| | Boiling Point | 234ºC
| | Flash Point | 79ºC
| | Refractive Index | 1.64-1.644
| | Water Solubility | INSOLUBLE
| | Stability | Stable at room temperature in closed containers under normal storage and handling conditions.
| | Storage Condition | Keep away from heat, sparks, and flame. Keep away from sources of ignition. Keep container closed when not in use. Store in a cool, dry, well-ventilated area away from incompatible substances. Corrosives area. Do not expose to air. Store under an inert atm
| | Vapor Pressure | 0.0517mmHg at 25°C |
2-Aminothiophenol is an organosulfur compound with the formula C6H4(SH)(NH2). It is a colorless oily solid, although impure samples can be deeply colored. It is soluble in organic solvents and in basic water. It is a precursor to benzothiazoles, some of which are bioactive or are dyes. Isomers of aminothiophenols include 3-aminothiophenol and 4-aminothiophenol.
2-Aminothiophenol can prepared in two steps, starting with the reaction of aniline with carbon disulfide followed by hydrolysis of the resulting mercaptobenzothiazole. It can also obtained by zinc reduction of 2-nitrobenzenesulfonyl chloride.
| | Appearance & Physical State | clear yellow to amber liquid
| | Density | 1.16
| | Melting Point | 23-26ºC
| | Boiling Point | 234ºC
| | Flash Point | 79ºC
| | Refractive Index | 1.64-1.644
| | Water Solubility | INSOLUBLE
| | Stability | Stable at room temperature in closed containers under normal storage and handling conditions.
| | Storage Condition | Keep away from heat, sparks, and flame. Keep away from sources of ignition. Keep container closed when not in use. Store in a cool, dry, well-ventilated area away from incompatible substances. Corrosives area. Do not expose to air. Store under an inert atm
| | Vapor Pressure | 0.0517mmHg at 25°C |
2-Aminothiophenol is an organosulfur compound with the formula C6H4(SH)(NH2). It is a colorless oily solid, although impure samples can be deeply colored. It is soluble in organic solvents and in basic water. It is a precursor to benzothiazoles, some of which are bioactive or are dyes. Isomers of aminothiophenols include 3-aminothiophenol and 4-aminothiophenol.
2-Aminothiophenol can prepared in two steps, starting with the reaction of aniline with carbon disulfide followed by hydrolysis of the resulting mercaptobenzothiazole. It can also obtained by zinc reduction of 2-nitrobenzenesulfonyl chloride.
| | Appearance & Physical State | clear yellow to amber liquid
| | Density | 1.16
| | Melting Point | 23-26ºC
| | Boiling Point | 234ºC
| | Flash Point | 79ºC
| | Refractive Index | 1.64-1.644
| | Water Solubility | INSOLUBLE
| | Stability | Stable at room temperature in closed containers under normal storage and handling conditions.
| | Storage Condition | Keep away from heat, sparks, and flame. Keep away from sources of ignition. Keep container closed when not in use. Store in a cool, dry, well-ventilated area away from incompatible substances. Corrosives area. Do not expose to air. Store under an inert atm
| | Vapor Pressure | 0.0517mmHg at 25°C |
2-Aminothiophenol is an organosulfur compound with the formula C6H4(SH)(NH2). It is a colorless oily solid, although impure samples can be deeply colored. It is soluble in organic solvents and in basic water. It is a precursor to benzothiazoles, some of which are bioactive or are dyes. Isomers of aminothiophenols include 3-aminothiophenol and 4-aminothiophenol.
2-Aminothiophenol can prepared in two steps, starting with the reaction of aniline with carbon disulfide followed by hydrolysis of the resulting mercaptobenzothiazole. It can also obtained by zinc reduction of 2-nitrobenzenesulfonyl chloride.
| | Appearance & Physical State | clear yellow to amber liquid
| | Density | 1.16
| | Melting Point | 23-26ºC
| | Boiling Point | 234ºC
| | Flash Point | 79ºC
| | Refractive Index | 1.64-1.644
| | Water Solubility | INSOLUBLE
| | Stability | Stable at room temperature in closed containers under normal storage and handling conditions.
| | Storage Condition | Keep away from heat, sparks, and flame. Keep away from sources of ignition. Keep container closed when not in use. Store in a cool, dry, well-ventilated area away from incompatible substances. Corrosives area. Do not expose to air. Store under an inert atm
| | Vapor Pressure | 0.0517mmHg at 25°C |
2-Aminothiophenol is an organosulfur compound with the formula C6H4(SH)(NH2). It is a colorless oily solid, although impure samples can be deeply colored. It is soluble in organic solvents and in basic water. It is a precursor to benzothiazoles, some of which are bioactive or are dyes. Isomers of aminothiophenols include 3-aminothiophenol and 4-aminothiophenol.
2-Aminothiophenol can prepared in two steps, starting with the reaction of aniline with carbon disulfide followed by hydrolysis of the resulting mercaptobenzothiazole. It can also obtained by zinc reduction of 2-nitrobenzenesulfonyl chloride.
| | Appearance & Physical State | clear yellow to amber liquid
| | Density | 1.16
| | Melting Point | 23-26ºC
| | Boiling Point | 234ºC
| | Flash Point | 79ºC
| | Refractive Index | 1.64-1.644
| | Water Solubility | INSOLUBLE
| | Stability | Stable at room temperature in closed containers under normal storage and handling conditions.
| | Storage Condition | Keep away from heat, sparks, and flame. Keep away from sources of ignition. Keep container closed when not in use. Store in a cool, dry, well-ventilated area away from incompatible substances. Corrosives area. Do not expose to air. Store under an inert atm
| | Vapor Pressure | 0.0517mmHg at 25°C |
2-Aminothiophenol is an organosulfur compound with the formula C6H4(SH)(NH2). It is a colorless oily solid, although impure samples can be deeply colored. It is soluble in organic solvents and in basic water. It is a precursor to benzothiazoles, some of which are bioactive or are dyes. Isomers of aminothiophenols include 3-aminothiophenol and 4-aminothiophenol.
2-Aminothiophenol can prepared in two steps, starting with the reaction of aniline with carbon disulfide followed by hydrolysis of the resulting mercaptobenzothiazole. It can also obtained by zinc reduction of 2-nitrobenzenesulfonyl chloride.
| | Appearance & Physical State | clear yellow to amber liquid
| | Density | 1.16
| | Melting Point | 23-26ºC
| | Boiling Point | 234ºC
| | Flash Point | 79ºC
| | Refractive Index | 1.64-1.644
| | Water Solubility | INSOLUBLE
| | Stability | Stable at room temperature in closed containers under normal storage and handling conditions.
| | Storage Condition | Keep away from heat, sparks, and flame. Keep away from sources of ignition. Keep container closed when not in use. Store in a cool, dry, well-ventilated area away from incompatible substances. Corrosives area. Do not expose to air. Store under an inert atm
| | Vapor Pressure | 0.0517mmHg at 25°C |
2-Aminothiophenol is an organosulfur compound with the formula C6H4(SH)(NH2). It is a colorless oily solid, although impure samples can be deeply colored. It is soluble in organic solvents and in basic water. It is a precursor to benzothiazoles, some of which are bioactive or are dyes. Isomers of aminothiophenols include 3-aminothiophenol and 4-aminothiophenol.
2-Aminothiophenol can prepared in two steps, starting with the reaction of aniline with carbon disulfide followed by hydrolysis of the resulting mercaptobenzothiazole. It can also obtained by zinc reduction of 2-nitrobenzenesulfonyl chloride.
| | Appearance & Physical State | clear yellow to amber liquid
| | Density | 1.16
| | Melting Point | 23-26ºC
| | Boiling Point | 234ºC
| | Flash Point | 79ºC
| | Refractive Index | 1.64-1.644
| | Water Solubility | INSOLUBLE
| | Stability | Stable at room temperature in closed containers under normal storage and handling conditions.
| | Storage Condition | Keep away from heat, sparks, and flame. Keep away from sources of ignition. Keep container closed when not in use. Store in a cool, dry, well-ventilated area away from incompatible substances. Corrosives area. Do not expose to air. Store under an inert atm
| | Vapor Pressure | 0.0517mmHg at 25°C |
2-Aminothiophenol is an organosulfur compound with the formula C6H4(SH)(NH2). It is a colorless oily solid, although impure samples can be deeply colored. It is soluble in organic solvents and in basic water. It is a precursor to benzothiazoles, some of which are bioactive or are dyes. Isomers of aminothiophenols include 3-aminothiophenol and 4-aminothiophenol.
2-Aminothiophenol can prepared in two steps, starting with the reaction of aniline with carbon disulfide followed by hydrolysis of the resulting mercaptobenzothiazole. It can also obtained by zinc reduction of 2-nitrobenzenesulfonyl chloride.
| | Appearance & Physical State | clear yellow to amber liquid
| | Density | 1.16
| | Melting Point | 23-26ºC
| | Boiling Point | 234ºC
| | Flash Point | 79ºC
| | Refractive Index | 1.64-1.644
| | Water Solubility | INSOLUBLE
| | Stability | Stable at room temperature in closed containers under normal storage and handling conditions.
| | Storage Condition | Keep away from heat, sparks, and flame. Keep away from sources of ignition. Keep container closed when not in use. Store in a cool, dry, well-ventilated area away from incompatible substances. Corrosives area. Do not expose to air. Store under an inert atm
| | Vapor Pressure | 0.0517mmHg at 25°C |
2-Aminothiophenol is an organosulfur compound with the formula C6H4(SH)(NH2). It is a colorless oily solid, although impure samples can be deeply colored. It is soluble in organic solvents and in basic water. It is a precursor to benzothiazoles, some of which are bioactive or are dyes. Isomers of aminothiophenols include 3-aminothiophenol and 4-aminothiophenol.
2-Aminothiophenol can prepared in two steps, starting with the reaction of aniline with carbon disulfide followed by hydrolysis of the resulting mercaptobenzothiazole. It can also obtained by zinc reduction of 2-nitrobenzenesulfonyl chloride.
| | Appearance & Physical State | clear yellow to amber liquid
| | Density | 1.16
| | Melting Point | 23-26ºC
| | Boiling Point | 234ºC
| | Flash Point | 79ºC
| | Refractive Index | 1.64-1.644
| | Water Solubility | INSOLUBLE
| | Stability | Stable at room temperature in closed containers under normal storage and handling conditions.
| | Storage Condition | Keep away from heat, sparks, and flame. Keep away from sources of ignition. Keep container closed when not in use. Store in a cool, dry, well-ventilated area away from incompatible substances. Corrosives area. Do not expose to air. Store under an inert atm
| | Vapor Pressure | 0.0517mmHg at 25°C |
2-Aminothiophenol is an organosulfur compound with the formula C6H4(SH)(NH2). It is a colorless oily solid, although impure samples can be deeply colored. It is soluble in organic solvents and in basic water. It is a precursor to benzothiazoles, some of which are bioactive or are dyes. Isomers of aminothiophenols include 3-aminothiophenol and 4-aminothiophenol.
2-Aminothiophenol can prepared in two steps, starting with the reaction of aniline with carbon disulfide followed by hydrolysis of the resulting mercaptobenzothiazole. It can also obtained by zinc reduction of 2-nitrobenzenesulfonyl chloride.
| | Appearance & Physical State | clear yellow to amber liquid
| | Density | 1.16
| | Melting Point | 23-26ºC
| | Boiling Point | 234ºC
| | Flash Point | 79ºC
| | Refractive Index | 1.64-1.644
| | Water Solubility | INSOLUBLE
| | Stability | Stable at room temperature in closed containers under normal storage and handling conditions.
| | Storage Condition | Keep away from heat, sparks, and flame. Keep away from sources of ignition. Keep container closed when not in use. Store in a cool, dry, well-ventilated area away from incompatible substances. Corrosives area. Do not expose to air. Store under an inert atm
| | Vapor Pressure | 0.0517mmHg at 25°C |
2-Aminothiophenol is an organosulfur compound with the formula C6H4(SH)(NH2). It is a colorless oily solid, although impure samples can be deeply colored. It is soluble in organic solvents and in basic water. It is a precursor to benzothiazoles, some of which are bioactive or are dyes. Isomers of aminothiophenols include 3-aminothiophenol and 4-aminothiophenol.
2-Aminothiophenol can prepared in two steps, starting with the reaction of aniline with carbon disulfide followed by hydrolysis of the resulting mercaptobenzothiazole. It can also obtained by zinc reduction of 2-nitrobenzenesulfonyl chloride.
| | Appearance & Physical State | clear yellow to amber liquid
| | Density | 1.16
| | Melting Point | 23-26ºC
| | Boiling Point | 234ºC
| | Flash Point | 79ºC
| | Refractive Index | 1.64-1.644
| | Water Solubility | INSOLUBLE
| | Stability | Stable at room temperature in closed containers under normal storage and handling conditions.
| | Storage Condition | Keep away from heat, sparks, and flame. Keep away from sources of ignition. Keep container closed when not in use. Store in a cool, dry, well-ventilated area away from incompatible substances. Corrosives area. Do not expose to air. Store under an inert atm
| | Vapor Pressure | 0.0517mmHg at 25°C |
2-Aminothiophenol is an organosulfur compound with the formula C6H4(SH)(NH2). It is a colorless oily solid, although impure samples can be deeply colored. It is soluble in organic solvents and in basic water. It is a precursor to benzothiazoles, some of which are bioactive or are dyes. Isomers of aminothiophenols include 3-aminothiophenol and 4-aminothiophenol.
2-Aminothiophenol can prepared in two steps, starting with the reaction of aniline with carbon disulfide followed by hydrolysis of the resulting mercaptobenzothiazole. It can also obtained by zinc reduction of 2-nitrobenzenesulfonyl chloride.
| | Appearance & Physical State | clear yellow to amber liquid
| | Density | 1.16
| | Melting Point | 23-26ºC
| | Boiling Point | 234ºC
| | Flash Point | 79ºC
| | Refractive Index | 1.64-1.644
| | Water Solubility | INSOLUBLE
| | Stability | Stable at room temperature in closed containers under normal storage and handling conditions.
| | Storage Condition | Keep away from heat, sparks, and flame. Keep away from sources of ignition. Keep container closed when not in use. Store in a cool, dry, well-ventilated area away from incompatible substances. Corrosives area. Do not expose to air. Store under an inert atm
| | Vapor Pressure | 0.0517mmHg at 25°C |
2-Aminothiophenol is an organosulfur compound with the formula C6H4(SH)(NH2). It is a colorless oily solid, although impure samples can be deeply colored. It is soluble in organic solvents and in basic water. It is a precursor to benzothiazoles, some of which are bioactive or are dyes. Isomers of aminothiophenols include 3-aminothiophenol and 4-aminothiophenol.
2-Aminothiophenol can prepared in two steps, starting with the reaction of aniline with carbon disulfide followed by hydrolysis of the resulting mercaptobenzothiazole. It can also obtained by zinc reduction of 2-nitrobenzenesulfonyl chloride.
| | Appearance & Physical State | clear yellow to amber liquid
| | Density | 1.16
| | Melting Point | 23-26ºC
| | Boiling Point | 234ºC
| | Flash Point | 79ºC
| | Refractive Index | 1.64-1.644
| | Water Solubility | INSOLUBLE
| | Stability | Stable at room temperature in closed containers under normal storage and handling conditions.
| | Storage Condition | Keep away from heat, sparks, and flame. Keep away from sources of ignition. Keep container closed when not in use. Store in a cool, dry, well-ventilated area away from incompatible substances. Corrosives area. Do not expose to air. Store under an inert atm
| | Vapor Pressure | 0.0517mmHg at 25°C |
2-Aminothiophenol is an organosulfur compound with the formula C6H4(SH)(NH2). It is a colorless oily solid, although impure samples can be deeply colored. It is soluble in organic solvents and in basic water. It is a precursor to benzothiazoles, some of which are bioactive or are dyes. Isomers of aminothiophenols include 3-aminothiophenol and 4-aminothiophenol.
2-Aminothiophenol can prepared in two steps, starting with the reaction of aniline with carbon disulfide followed by hydrolysis of the resulting mercaptobenzothiazole. It can also obtained by zinc reduction of 2-nitrobenzenesulfonyl chloride.
| | Appearance & Physical State | clear yellow to amber liquid
| | Density | 1.16
| | Melting Point | 23-26ºC
| | Boiling Point | 234ºC
| | Flash Point | 79ºC
| | Refractive Index | 1.64-1.644
| | Water Solubility | INSOLUBLE
| | Stability | Stable at room temperature in closed containers under normal storage and handling conditions.
| | Storage Condition | Keep away from heat, sparks, and flame. Keep away from sources of ignition. Keep container closed when not in use. Store in a cool, dry, well-ventilated area away from incompatible substances. Corrosives area. Do not expose to air. Store under an inert atm
| | Vapor Pressure | 0.0517mmHg at 25°C |
|
|
|
Privileged suppliers
Last update 2024-04-26
|


 details
details
_Co_Ltd_1_575.jpg)







