|
|
|

|
Suppliers for
Zanamivir hydrate
|
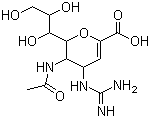
Properties | | CAS |
139110-80-8 | | Formula |
C12H20N4O7 |
|
|
4 Registered suppliers
More details are to be found here
Description : Zanamivir is a neuraminidase inhibitor used in the treatment and prophylaxis of influenza caused by influenza A virus and influenza B virus. - Molecular Weight :350.33
- Melting Point :> 245 °C (dec.)
- Purity :> 98%
Molecular Formula : C12H22N4O8 Canonical SMILES : CC(=O)NC1C(C=C(OC1C(C(CO)O)O)C(=O)O)N=C(N)N InChI : InChI=1S/C12H20N4O7/c1-4(18)15-8-5(16-12(13)14)2-7(11(21)22)23-10(8)9(20)6(19)3-17/h2,5-6,8-10,17,19-20H,3H2,1H3,(H,15,18)(H,21,22)(H4,13,14,16)/t5-,6+,8+,9+,10+/m0/s1
InChIKey :
ARAIBEBZBOPLMB-UFGQHTETSA-N
Solubility :
In vitro: 10 mM in DMSO
Appearance :
White Solid
Storage : Powder: -20°C: 3 years 4°C: 2 years In solvent: -80°C: 6 months -20°C: 1 month
Synonyms :
Relenza; GG 167; 2,3-Didehydro-2,4-dideoxy-4-guanidine-N-acetyl-acetyl-D-neuraminic acid; 4-Guanidino-2,4-dideoxy-2,3-didehydro-N-acetylneuraminic acid; 5-Acetamino-2,6-anhydro-4-guanidino-3,4,5-trideoxy-D-galcto-non-enoic acid
More details are to be found here
Description : Zanamivir is a neuraminidase inhibitor used in the treatment and prophylaxis of influenza caused by influenza A virus and influenza B virus. - Molecular Weight :350.33
- Melting Point :> 245 °C (dec.)
- Purity :> 98%
Molecular Formula : C12H22N4O8 Canonical SMILES : CC(=O)NC1C(C=C(OC1C(C(CO)O)O)C(=O)O)N=C(N)N InChI : InChI=1S/C12H20N4O7/c1-4(18)15-8-5(16-12(13)14)2-7(11(21)22)23-10(8)9(20)6(19)3-17/h2,5-6,8-10,17,19-20H,3H2,1H3,(H,15,18)(H,21,22)(H4,13,14,16)/t5-,6+,8+,9+,10+/m0/s1
InChIKey :
ARAIBEBZBOPLMB-UFGQHTETSA-N
Solubility :
In vitro: 10 mM in DMSO
Appearance :
White Solid
Storage : Powder: -20°C: 3 years 4°C: 2 years In solvent: -80°C: 6 months -20°C: 1 month
Synonyms :
Relenza; GG 167; 2,3-Didehydro-2,4-dideoxy-4-guanidine-N-acetyl-acetyl-D-neuraminic acid; 4-Guanidino-2,4-dideoxy-2,3-didehydro-N-acetylneuraminic acid; 5-Acetamino-2,6-anhydro-4-guanidino-3,4,5-trideoxy-D-galcto-non-enoic acid
More details are to be found here
|
|
|
Privileged suppliers
Last update 2024-04-26
|


 details
details details
details


