|
|
|

|
Suppliers for
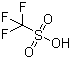
Trifluoromethanesulfonic acid
|
Properties | | CAS |
1493-13-6 | | Formula |
CHF3O3S | | EINECS |
216-087-5 |
|
|
40 Registered suppliers
Specification
| Appearance Colorless to light yellow transparent liquid
| Purity 99% min
| Free Fluorine (ppm) 10 max
| | Moisture (ppm) 500 max |
Specification
| Appearance Colorless to light yellow transparent liquid
| Purity 99% min
| Free Fluorine (ppm) 10 max
| | Moisture (ppm) 500 max |
More details are to be found here
Molecular Formula: CHF3O3S Molecular Weight: 150.0770496 Hazard Symbols: C UN Number: UN 3265 8/PG 2 WGKGermany: 2 PackingGroup: II HS Code: 29049020
More details are to be found here
Molecular Formula: CHF3O3S Molecular Weight: 150.0770496 Hazard Symbols: C UN Number: UN 3265 8/PG 2 WGKGermany: 2 PackingGroup: II HS Code: 29049020
More details are to be found here
Molecular Formula: CHF3O3S Molecular Weight: 150.0770496 Hazard Symbols: C UN Number: UN 3265 8/PG 2 WGKGermany: 2 PackingGroup: II HS Code: 29049020
More details are to be found here
Molecular Formula: CHF3O3S Molecular Weight: 150.0770496 Hazard Symbols: C UN Number: UN 3265 8/PG 2 WGKGermany: 2 PackingGroup: II HS Code: 29049020
More details are to be found here
Molecular Formula: CHF3O3S Molecular Weight: 150.0770496 Hazard Symbols: C UN Number: UN 3265 8/PG 2 WGKGermany: 2 PackingGroup: II HS Code: 29049020
More details are to be found here
| MF | CHF3O3S
| | EINECS No. | 216-087-5
| | Place of Origin | China
| | Type | Syntheses Material Intermediates
| | Purity | 99.5%min
| | Brand Name | Unilong |
EINECS :216-087-5
Molecular formula :CHF3O3S
EINECS :216-087-5
Molecular formula :CHF3O3S
EINECS :216-087-5
Molecular formula :CHF3O3S
EINECS :216-087-5
Molecular formula :CHF3O3S
EINECS :216-087-5
Molecular formula :CHF3O3S
EINECS :216-087-5
Molecular formula :CHF3O3S
EINECS :216-087-5
Molecular formula :CHF3O3S
EINECS :216-087-5
Molecular formula :CHF3O3S
| Appearance | colorless to light yellow liquid
| | Purity | 99.5% min |
| Appearance | colorless to light yellow liquid
| | Purity | 99.5% min |
| Appearance | colorless to light yellow liquid
| | Purity | 99.5% min |
| Appearance | colorless to light yellow liquid
| | Purity | 99.5% min |
| Appearance | colorless to light yellow liquid
| | Purity | 99.5% min |
English name: trifluoromethanesulfonic acid
Trifluoromethanesulfonic acid; trifluoromethanesulfonic acid; trifluoromethanesulfonic acid; trifluoromethanesulfonic acid; trifluoromethanesulfonic acid; trifluoromethanesulfonic acid, 98 +%; trifluoroacetic acid; trifluoromethanesulfonic acid; Trifluoromethanesulfonic acid (colorless transparent liquid)
English name: Trifluoromethanesulfonic acid
Trifluoromethanesulfonic; trifluoro-methanesulfonicaci; PERFLUOROMETHANESULFONIC ACID; PFC-MS; TRIFLUOROMETHANESULPHONIC ACID; TRIFLUOROMETHANESULFONIC ACID;
CAS: 1493-13-6
Molecular formula: CHF3O3S
Molecular weight: 150.08
EINECS: 216-087-5
Melting point -40 ° C
Boiling Point: 162 ° C (lit.)
Density 1.696 g / mL at 25 ° C (lit.)
Vapor density 5.2 (vs air)
Vapor pressure 8 mm Hg (25 ° C)
Refractive index n20 / D 1.327 (lit.)
Flash point None
Storage conditions -20 ° C
Color slightly brown
SOLUBLE WATER SOLUBLE
Sensitivity
Merck 9676
BRN 1812100
Stable. Incompatible with acids, alkalis, metals.
CAS database 1493-13-6 (CAS DataBase Reference)
NIST chemical substance information CF3SO3H (1493-13-6)
Preparation: yellow-brown liquid. The boiling point of 167 ~ 170 ℃. Refractive index 1.331. The relative density of 1.708. As the strongest organic acids, easily soluble in water.
To carbon disulfide as raw materials, and iodine pentafluoride reaction of trifluoromethyl disulfide. And mercury reaction trifluoromethyl mercaptan (CF3S) 2Hg, which after oxidation of hydrogen fluoride that was trifluoromethanesulfonic acid.
Use For organic synthesis
Uses a wide range of uses, is known as one of the most powerful organic acids, is a universal synthesis tool. With strong corrosive, hygroscopicity, widely used in medicine, chemical and other industries. Such as nucleosides, antibiotics, steroids, proteins, glycosides, vitamin synthesis, silicone rubber modification.
Applications Isomerization and alkylation of catalysts. Preparation of 2,3-dihydro-2-indanone, tetralone. Glycoside removal from glycoproteins.
English name: trifluoromethanesulfonic acid
Trifluoromethanesulfonic acid; trifluoromethanesulfonic acid; trifluoromethanesulfonic acid; trifluoromethanesulfonic acid; trifluoromethanesulfonic acid; trifluoromethanesulfonic acid, 98 +%; trifluoroacetic acid; trifluoromethanesulfonic acid; Trifluoromethanesulfonic acid (colorless transparent liquid)
English name: Trifluoromethanesulfonic acid
Trifluoromethanesulfonic; trifluoro-methanesulfonicaci; PERFLUOROMETHANESULFONIC ACID; PFC-MS; TRIFLUOROMETHANESULPHONIC ACID; TRIFLUOROMETHANESULFONIC ACID;
CAS: 1493-13-6
Molecular formula: CHF3O3S
Molecular weight: 150.08
EINECS: 216-087-5
Melting point -40 ° C
Boiling Point: 162 ° C (lit.)
Density 1.696 g / mL at 25 ° C (lit.)
Vapor density 5.2 (vs air)
Vapor pressure 8 mm Hg (25 ° C)
Refractive index n20 / D 1.327 (lit.)
Flash point None
Storage conditions -20 ° C
Color slightly brown
SOLUBLE WATER SOLUBLE
Sensitivity
Merck 9676
BRN 1812100
Stable. Incompatible with acids, alkalis, metals.
CAS database 1493-13-6 (CAS DataBase Reference)
NIST chemical substance information CF3SO3H (1493-13-6)
Preparation: yellow-brown liquid. The boiling point of 167 ~ 170 ℃. Refractive index 1.331. The relative density of 1.708. As the strongest organic acids, easily soluble in water.
To carbon disulfide as raw materials, and iodine pentafluoride reaction of trifluoromethyl disulfide. And mercury reaction trifluoromethyl mercaptan (CF3S) 2Hg, which after oxidation of hydrogen fluoride that was trifluoromethanesulfonic acid.
Use For organic synthesis
Uses a wide range of uses, is known as one of the most powerful organic acids, is a universal synthesis tool. With strong corrosive, hygroscopicity, widely used in medicine, chemical and other industries. Such as nucleosides, antibiotics, steroids, proteins, glycosides, vitamin synthesis, silicone rubber modification.
Applications Isomerization and alkylation of catalysts. Preparation of 2,3-dihydro-2-indanone, tetralone. Glycoside removal from glycoproteins.
English name: trifluoromethanesulfonic acid
Trifluoromethanesulfonic acid; trifluoromethanesulfonic acid; trifluoromethanesulfonic acid; trifluoromethanesulfonic acid; trifluoromethanesulfonic acid; trifluoromethanesulfonic acid, 98 +%; trifluoroacetic acid; trifluoromethanesulfonic acid; Trifluoromethanesulfonic acid (colorless transparent liquid)
English name: Trifluoromethanesulfonic acid
Trifluoromethanesulfonic; trifluoro-methanesulfonicaci; PERFLUOROMETHANESULFONIC ACID; PFC-MS; TRIFLUOROMETHANESULPHONIC ACID; TRIFLUOROMETHANESULFONIC ACID;
CAS: 1493-13-6
Molecular formula: CHF3O3S
Molecular weight: 150.08
EINECS: 216-087-5
Melting point -40 ° C
Boiling Point: 162 ° C (lit.)
Density 1.696 g / mL at 25 ° C (lit.)
Vapor density 5.2 (vs air)
Vapor pressure 8 mm Hg (25 ° C)
Refractive index n20 / D 1.327 (lit.)
Flash point None
Storage conditions -20 ° C
Color slightly brown
SOLUBLE WATER SOLUBLE
Sensitivity
Merck 9676
BRN 1812100
Stable. Incompatible with acids, alkalis, metals.
CAS database 1493-13-6 (CAS DataBase Reference)
NIST chemical substance information CF3SO3H (1493-13-6)
Preparation: yellow-brown liquid. The boiling point of 167 ~ 170 ℃. Refractive index 1.331. The relative density of 1.708. As the strongest organic acids, easily soluble in water.
To carbon disulfide as raw materials, and iodine pentafluoride reaction of trifluoromethyl disulfide. And mercury reaction trifluoromethyl mercaptan (CF3S) 2Hg, which after oxidation of hydrogen fluoride that was trifluoromethanesulfonic acid.
Use For organic synthesis
Uses a wide range of uses, is known as one of the most powerful organic acids, is a universal synthesis tool. With strong corrosive, hygroscopicity, widely used in medicine, chemical and other industries. Such as nucleosides, antibiotics, steroids, proteins, glycosides, vitamin synthesis, silicone rubber modification.
Applications Isomerization and alkylation of catalysts. Preparation of 2,3-dihydro-2-indanone, tetralone. Glycoside removal from glycoproteins.
English name: trifluoromethanesulfonic acid
Trifluoromethanesulfonic acid; trifluoromethanesulfonic acid; trifluoromethanesulfonic acid; trifluoromethanesulfonic acid; trifluoromethanesulfonic acid; trifluoromethanesulfonic acid, 98 +%; trifluoroacetic acid; trifluoromethanesulfonic acid; Trifluoromethanesulfonic acid (colorless transparent liquid)
English name: Trifluoromethanesulfonic acid
Trifluoromethanesulfonic; trifluoro-methanesulfonicaci; PERFLUOROMETHANESULFONIC ACID; PFC-MS; TRIFLUOROMETHANESULPHONIC ACID; TRIFLUOROMETHANESULFONIC ACID;
CAS: 1493-13-6
Molecular formula: CHF3O3S
Molecular weight: 150.08
EINECS: 216-087-5
Melting point -40 ° C
Boiling Point: 162 ° C (lit.)
Density 1.696 g / mL at 25 ° C (lit.)
Vapor density 5.2 (vs air)
Vapor pressure 8 mm Hg (25 ° C)
Refractive index n20 / D 1.327 (lit.)
Flash point None
Storage conditions -20 ° C
Color slightly brown
SOLUBLE WATER SOLUBLE
Sensitivity
Merck 9676
BRN 1812100
Stable. Incompatible with acids, alkalis, metals.
CAS database 1493-13-6 (CAS DataBase Reference)
NIST chemical substance information CF3SO3H (1493-13-6)
Preparation: yellow-brown liquid. The boiling point of 167 ~ 170 ℃. Refractive index 1.331. The relative density of 1.708. As the strongest organic acids, easily soluble in water.
To carbon disulfide as raw materials, and iodine pentafluoride reaction of trifluoromethyl disulfide. And mercury reaction trifluoromethyl mercaptan (CF3S) 2Hg, which after oxidation of hydrogen fluoride that was trifluoromethanesulfonic acid.
Use For organic synthesis
Uses a wide range of uses, is known as one of the most powerful organic acids, is a universal synthesis tool. With strong corrosive, hygroscopicity, widely used in medicine, chemical and other industries. Such as nucleosides, antibiotics, steroids, proteins, glycosides, vitamin synthesis, silicone rubber modification.
Applications Isomerization and alkylation of catalysts. Preparation of 2,3-dihydro-2-indanone, tetralone. Glycoside removal from glycoproteins.
English name: trifluoromethanesulfonic acid
Trifluoromethanesulfonic acid; trifluoromethanesulfonic acid; trifluoromethanesulfonic acid; trifluoromethanesulfonic acid; trifluoromethanesulfonic acid; trifluoromethanesulfonic acid, 98 +%; trifluoroacetic acid; trifluoromethanesulfonic acid; Trifluoromethanesulfonic acid (colorless transparent liquid)
English name: Trifluoromethanesulfonic acid
Trifluoromethanesulfonic; trifluoro-methanesulfonicaci; PERFLUOROMETHANESULFONIC ACID; PFC-MS; TRIFLUOROMETHANESULPHONIC ACID; TRIFLUOROMETHANESULFONIC ACID;
CAS: 1493-13-6
Molecular formula: CHF3O3S
Molecular weight: 150.08
EINECS: 216-087-5
Melting point -40 ° C
Boiling Point: 162 ° C (lit.)
Density 1.696 g / mL at 25 ° C (lit.)
Vapor density 5.2 (vs air)
Vapor pressure 8 mm Hg (25 ° C)
Refractive index n20 / D 1.327 (lit.)
Flash point None
Storage conditions -20 ° C
Color slightly brown
SOLUBLE WATER SOLUBLE
Sensitivity
Merck 9676
BRN 1812100
Stable. Incompatible with acids, alkalis, metals.
CAS database 1493-13-6 (CAS DataBase Reference)
NIST chemical substance information CF3SO3H (1493-13-6)
Preparation: yellow-brown liquid. The boiling point of 167 ~ 170 ℃. Refractive index 1.331. The relative density of 1.708. As the strongest organic acids, easily soluble in water.
To carbon disulfide as raw materials, and iodine pentafluoride reaction of trifluoromethyl disulfide. And mercury reaction trifluoromethyl mercaptan (CF3S) 2Hg, which after oxidation of hydrogen fluoride that was trifluoromethanesulfonic acid.
Use For organic synthesis
Uses a wide range of uses, is known as one of the most powerful organic acids, is a universal synthesis tool. With strong corrosive, hygroscopicity, widely used in medicine, chemical and other industries. Such as nucleosides, antibiotics, steroids, proteins, glycosides, vitamin synthesis, silicone rubber modification.
Applications Isomerization and alkylation of catalysts. Preparation of 2,3-dihydro-2-indanone, tetralone. Glycoside removal from glycoproteins.
| Product Specification | ≥ 99.5% |
| Product Specification | ≥ 99.5% |
| Product Specification | ≥ 99.5% |
| Product Specification | ≥ 99.5% |
| Product Specification | ≥ 99.5% |
| Product Specification | ≥ 99.5% |
| Product Specification | ≥ 99.5% |
|
|
|
Privileged suppliers
Last update 2024-04-30
|


 details
details details
details details
details








