|
|
|

|
Suppliers for
Calcium folinate
|
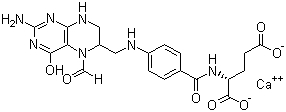
Properties | | CAS |
1492-18-8 | | Formula |
C20H21N7O7Ca | | EINECS |
216-082-8 |
|
|
19 Registered suppliers
Molecular Formula: C20H21N7O7.Ca Molecular Weight: 511.51
More details are to be found here
Molecular Formula: C20H21N7O7.Ca Molecular Weight: 511.51
More details are to be found here
Molecular Formula: C20H21N7O7.Ca Molecular Weight: 511.51
More details are to be found here
EINECS :216-082-8
Molecular formula :C20H21N7O7.Ca
EINECS :216-082-8
Molecular formula :C20H21N7O7.Ca
EINECS :216-082-8
Molecular formula :C20H21N7O7.Ca
EINECS :216-082-8
Molecular formula :C20H21N7O7.Ca
EINECS :216-082-8
Molecular formula :C20H21N7O7.Ca
More details are to be found here
More details are to be found here
More details are to be found here
More details are to be found here
More details are to be found here
Appearance:White or light yellow, amorphous or crystalline powder
Content: 97.0% ~ 102.0%
Use: one kind folic acid antidote, API
Peculiarity:Medicine Grade
Packing: 1kg/bag or 25kgs/drum
Min.Order Quantity:100g
Grade Standard:Medicine Grade
Product Specifications:
1.PH:6.0-8.0
2.Heavy metals:NMT 0.005%
3.Water Content:NMT 17.0%
4.Calcium:7.54%~8.14%
5.Standard: USP31
Calcium folinate/leucovorin calcium/Folinic acid is administered at the appropriate time
following methotrexate as part of a total chemotherapeutic plan, where it may `rescue`
bone marrow and gastrointestinal mucosa cells from methotrexate.
There is no apparent effect on preexisting methotrexate-induced nephrotoxicity.
Appearance:White or light yellow, amorphous or crystalline powder
Content: 97.0% ~ 102.0%
Use: one kind folic acid antidote, API
Peculiarity:Medicine Grade
Packing: 1kg/bag or 25kgs/drum
Min.Order Quantity:100g
Grade Standard:Medicine Grade
Product Specifications:
1.PH:6.0-8.0
2.Heavy metals:NMT 0.005%
3.Water Content:NMT 17.0%
4.Calcium:7.54%~8.14%
5.Standard: USP31
Calcium folinate/leucovorin calcium/Folinic acid is administered at the appropriate time
following methotrexate as part of a total chemotherapeutic plan, where it may `rescue`
bone marrow and gastrointestinal mucosa cells from methotrexate.
There is no apparent effect on preexisting methotrexate-induced nephrotoxicity.
|
|
|
Privileged suppliers
Last update 2024-06-14
|


 details
details details
details








