|
|
|

|
Suppliers for
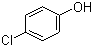
p-Chlorophenol
|
Properties | | CAS |
106-48-9 | | Formula |
C6H5ClO | | EINECS |
203-402-6 |
|
|
11 Registered suppliers
| Appearance | White flake crystalloid
| | Melting point | 39-43℃
| | Water | ≤ 0.5%
| | Assay | ≥ 99.0% |
| Product Name | 4-Chlorophenol
| | Synonyms | 4-chloro-1-hydroxybenzene; 4-chloro-pheno;4-Chlorophenol; chlorophenols,solid; p-Chlorfenol
| | CAS | 106-48-9
| | Product Categories | Aromatic Phenols;Organics; Chloride chemicals;Alpha sort; C; C Alphabetic; CH; Pesticides&Metabolites; Volatiles/ Semivolatiles; Organic Building Blocks; Oxygen Compounds; Phenols; PhenolsAnalytical Reagents; Waste Water; Water Test; Oxygen Compounds;Potable- &; Waste Water; Waste-Water Testing;
| Chemical Properties
| | mp | 40-45 °C(lit.)
| | bp | 220 °C(lit.)
| | density | 1.306 g/mL at 25 °C(lit.)
| | vapor pressure | 1 mm Hg ( 49.8 °C)
| | refractive index | 1.5579
| | Fp | 240 °F
| | storage temp. | 0-6°C
| | Water solubility | 2.7 g/100 mL (20 ºC)
| | Sensitive | Air Sensitive
| | Stability | Stable. Incompatible with acid chlorides, acid anhydrides, oxidizing agents, iron.
|
| Safety Information
| | Hazard codes | Xn,N
| | Risk statements | 20/21/22-51/53-50
| | safety statements | 28-61-28A-24/25-60
| | Chemical Properties | off-white to light tan crystals or powder
| | Usage | Intermediates of Liquid Crystals
| | General Description White crystals with a strong phenol odor Slightly soluble to soluble in water, depending on the isomer, and denser than water. Noncombustible. Used as an intermediate in organic synthesis of dyes and drugs. |
Molecular Formula: C6H5ClO
Molecular Weight: 128.5563
Molecular Formula: C6H5ClO
Molecular Weight: 128.5563
Molecular Formula: C6H5ClO
Molecular Weight: 128.5563
Molecular Formula: C6H5ClO
Molecular Weight: 128.5563
Molecular Formula: C6H5ClO
Molecular Weight: 128.5563
Molecular Formula: C6H5ClO
Molecular Weight: 128.5563
1.Product Name: 4-Chlorophenol
2.Synonyms: 4-chloro-1-hydroxybenzene;4-chloro-pheno;4-Chlorophenol(form2);4-chloro-phenole;Applied 3-78;applied3-78;chlorophenols,solid;p-Chlorfenol
3.CAS: 106-48-9
4.MF: C6H5ClO
5.MW: 128.56
6.EINECS: 203-402-6
7.The FOB Shanghai price: 2045 USD/MT.
8.The package: 200kg/drum,80 drums for 16MT(one container).
9.The application:
4-Chlorophenol Usage And Synthesis
Chemical Properties off-white to light tan crystals or powder
Usage Intermediates of Liquid Crystals
Usage antineoplastic
General Description White crystals with a strong phenol odor Slightly soluble to soluble in water, depending on the isomer, and denser than water. Noncombustible. Used as an intermediate in organic synthesis of dyes and drugs.
Air & Water Reactions Slightly soluble to soluble in water.
Reactivity Profile CHLOROPHENOLS, SOLID are incompatible with acid chlorides, acid anhydrides and oxidizing agents. Also incompatible with iron . Liquefy and darken in color at temperatures above 108آ°F.
Health Hazard Inhalation causes headache, dizziness, weak pulse. Ingestion causes irritation of mouth and stomach; headache, dizziness, weak pulse. Contact with eyes causes severe irritation and burning. Contact with skin causes irritation and burn; if absorbed, causes same symptoms as inhalation.
Fire Hazard Special Hazards of Combustion Products: Toxic and irritating hydrogen chloride and chlorine gases may form in fires.
1.Product Name: 4-Chlorophenol
2.Synonyms: 4-chloro-1-hydroxybenzene;4-chloro-pheno;4-Chlorophenol(form2);4-chloro-phenole;Applied 3-78;applied3-78;chlorophenols,solid;p-Chlorfenol
3.CAS: 106-48-9
4.MF: C6H5ClO
5.MW: 128.56
6.EINECS: 203-402-6
7.The FOB Shanghai price: 2045 USD/MT.
8.The package: 200kg/drum,80 drums for 16MT(one container).
9.The application:
4-Chlorophenol Usage And Synthesis
Chemical Properties off-white to light tan crystals or powder
Usage Intermediates of Liquid Crystals
Usage antineoplastic
General Description White crystals with a strong phenol odor Slightly soluble to soluble in water, depending on the isomer, and denser than water. Noncombustible. Used as an intermediate in organic synthesis of dyes and drugs.
Air & Water Reactions Slightly soluble to soluble in water.
Reactivity Profile CHLOROPHENOLS, SOLID are incompatible with acid chlorides, acid anhydrides and oxidizing agents. Also incompatible with iron . Liquefy and darken in color at temperatures above 108آ°F.
Health Hazard Inhalation causes headache, dizziness, weak pulse. Ingestion causes irritation of mouth and stomach; headache, dizziness, weak pulse. Contact with eyes causes severe irritation and burning. Contact with skin causes irritation and burn; if absorbed, causes same symptoms as inhalation.
Fire Hazard Special Hazards of Combustion Products: Toxic and irritating hydrogen chloride and chlorine gases may form in fires.
1.Product Name: 4-Chlorophenol
2.Synonyms: 4-chloro-1-hydroxybenzene;4-chloro-pheno;4-Chlorophenol(form2);4-chloro-phenole;Applied 3-78;applied3-78;chlorophenols,solid;p-Chlorfenol
3.CAS: 106-48-9
4.MF: C6H5ClO
5.MW: 128.56
6.EINECS: 203-402-6
7.The FOB Shanghai price: 2045 USD/MT.
8.The package: 200kg/drum,80 drums for 16MT(one container).
9.The application:
4-Chlorophenol Usage And Synthesis
Chemical Properties off-white to light tan crystals or powder
Usage Intermediates of Liquid Crystals
Usage antineoplastic
General Description White crystals with a strong phenol odor Slightly soluble to soluble in water, depending on the isomer, and denser than water. Noncombustible. Used as an intermediate in organic synthesis of dyes and drugs.
Air & Water Reactions Slightly soluble to soluble in water.
Reactivity Profile CHLOROPHENOLS, SOLID are incompatible with acid chlorides, acid anhydrides and oxidizing agents. Also incompatible with iron . Liquefy and darken in color at temperatures above 108آ°F.
Health Hazard Inhalation causes headache, dizziness, weak pulse. Ingestion causes irritation of mouth and stomach; headache, dizziness, weak pulse. Contact with eyes causes severe irritation and burning. Contact with skin causes irritation and burn; if absorbed, causes same symptoms as inhalation.
Fire Hazard Special Hazards of Combustion Products: Toxic and irritating hydrogen chloride and chlorine gases may form in fires.
|
|
|
Privileged suppliers
Last update 2024-06-14
|


 details
details details
details details
details







