|
Proveedores de CAS
124-41-4
|
Identificación | | CAS |
124-41-4 | | Formula |
CH3ONa | | EINECS |
204-699-5 |
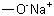
|
|
28 Proveedores registrados
Simagchem Corporation
P.R.China
Aecochem Corp.
P.R.China
Dayang Chem (Hangzhou) Co.,Ltd.
P.R.China
Sancai Industry Co.,Ltd.
P.R.China
| Appearance | Colorless and transparent viscous liquid
| | Content (total alkali) | 29.5%-31.0%
| | Sodium methoxide(NaOCH3) | 28.9-31.0%
| | Sodium hydroxide(NaOH) | ≤ 0.5%
| | Sodium carbonate(Na2CO3) | ≤ 0.1%
| | Moisture(H2O) | ≤ 0.2%
| | Methanol | 69.0-70.7% |
Hangzhou Keying Chem Co., Ltd.
P.R.China
Tianjin Zhongxin Chemtech Co., Ltd.
P.R.China
| Synonyms: | Methanol, sodium salt; Methanol,sodiumsalt; methoxysodium;
metilatosodico; sodiummethoxide, 30%(ww)solution in methanol
| | CAS: | 124-41-4
| | MF: | CH3NaO
| | MW: | 54.02
| | EINECS: | 204-699-5
| | Product Categories: | Organics;Classes of Metal Compounds;Na (Sodium) Compounds (excluding simple sodium salts);Typical Metal Compounds;metal alkoxide
| | Mol File: | 124-41-4.mol
| | mp : | -98 °C
| | bp : | 65 °C
| | density: | 0.97 g/mL at 20 °C
| | vapor density : | 1.1 (vs air)
| | vapor pressure : | 50 mm Hg ( 20 °C)
| | refractive index : | 1.3700
| | Fp : | 11 °C
| | storage temp. : | Flammables area
| | Water Solubility : | reacts
| | Sensitive : | Moisture Sensitive
| | Merck : | 14,8643
| | BRN : | 3592982
| | Stability: | Stability Highly flammable. Reacts violently with water. Keep container dry. Incompatible with water, acids, chlorinated solvents.
| | NIST: | Chemistry Reference Sodium methoxide(124-41-4)
| | Hazard Codes : | F,T,C
| | Risk Statements : | 11-23/24/25-34-39/23/24/25-36/38-14-36/37/38
| | Safety Statements : | 8-16-26-43-45-7-36/37-7/8-36/37/39
| | RIDADR: | UN 1431 4.2/PG 2
| | WGK Germany : | 1
| | F : | 9-21
| | TSCA : | Yes
| | HazardClass : | 4.2
| | PackingGroup : | II
| | Hazardous Substances Data: | 124-41-4(Hazardous Substances Data)
| General Description A colorless cloudy white liquid consisting of sodium methylate, a solid, dissolved in methyl alcohol. Density 9.0 lb / gal. Corrosive to metals and tissue. Used to process edible fats and oils and to make other chemicals.
| Air & Water Reactions Highly flammable. Ignites in moist air [Wischmeyer 1966]. Reacts with water to produce a mixed solution of sodium hydroxide and methyl alcohol.
| Reactivity Profile SODIUM METHYLATE is a strong base. Reacts with light metals forming H2 gas, with fire and explosion hazards. Too rapid addition of sodium methylate to a mixture of chloroform and methanol initiated an uncontrolled exothermic reaction between the chloroform and the methylate that caused a violent explosion [MCA Case History 693 1961]. Sodium methoxide is incompatible with 4-chloronitrobenzene and fluorinated cyclopropenyl methyl ethers, such as perfluoromethoxycyclopropene. The reactions are vigorous and may initiate ignition [Bretherick, 1995, pg. 191].
| Health Hazard TOXIC; inhalation, ingestion or skin contact with material may cause severe injury or death. Contact with molten substance may cause severe burns to skin and eyes. Avoid any skin contact. Effects of contact or inhalation may be delayed. Fire may produce irritating, corrosive and/or toxic gases. Runoff from fire control or dilution water may be corrosive and/or toxic and cause pollution.
| | Fire Hazard Non-combustible, substance itself does not burn but may decompose upon heating to produce corrosive and/or toxic fumes. Some are oxidizers and may ignite combustibles (wood, paper, oil, clothing, etc.). Contact with metals may evolve flammable hydrogen gas. Containers may explode when heated. |
| Product Name | Sodium methanolate
| | Synonyms: | Methanol, sodium salt; Methanol,sodiumsalt; methoxysodium;
| | CAS: | 124-41-4
| | MF: | CH3NaO
| | MW: | 54.02
| | EINECS: | 204-699-5
| | Product Categories: | Organics;Classes of Metal Compounds; Na (Sodium) Compounds (excluding simple sodium salts); Typical Metal Compounds;metal alkoxide
| | Mol File: | 124-41-4.mol
| | mp : | -98 °C
| | bp : | 65 °C
| | density: | 0.97 g/mL at 20 °C
| | vapor density : | 1.1 (vs air)
| | vapor pressure : | 50 mm Hg ( 20 °C)
| | refractive index : | 1.3700
| | Fp : | 11 °C
| | storage temp. : | Flammables area
| | Water Solubility : | reacts
| | Sensitive : | Moisture Sensitive
| | Merck : | 14,8643
| | BRN : | 3592982
| | Stability: | Stability Highly flammable. Reacts violently with water. Keep container dry. Incompatible with water, acids, chlorinated solvents.
| | Hazard Codes: | F,T,C
| | Risk Statements: | 11-23/24/25-34-39/23/24/25-36/38-14-36/37/38
| | Safety Statements : | 8-16-26-43-45-7-36/37-7/8-36/37/39
| | RIDADR : | UN 1431 4.2/PG 2
| | WGK Germany : | 1
| | F : | 9-21
| | TSCA : | Yes
| | HazardClass : | 4.2
| | PackingGroup : | II
| | Chemical Properties: | white powder
| | General Description A colorless cloudy white liquid consisting of sodium methylate, a solid, dissolved in methyl alcohol. Density 9.0 lb / gal. Corrosive to metals and tissue. Used to process edible fats and oils and to make other chemicals.
| | Air & Water Reactions Highly flammable. Ignites in moist air [Wischmeyer 1966]. Reacts with water to produce a mixed solution of sodium hydroxide and methyl alcohol.
| | Reactivity Profile SODIUM METHYLATE is a strong base. Reacts with light metals forming H2 gas, with fire and explosion hazards. Too rapid addition of sodium methylate to a mixture of chloroform and methanol initiated an uncontrolled exothermic reaction between the chloroform and the methylate that caused a violent explosion [MCA Case History 693 1961]. Sodium methoxide is incompatible with 4-chloronitrobenzene and fluorinated cyclopropenyl methyl ethers, such as perfluoromethoxycyclopropene. The reactions are vigorous and may initiate ignition [Bretherick, 1995, pg. 191].
| | Health Hazard TOXIC; inhalation, ingestion or skin contact with material may cause severe injury or death. Contact with molten substance may cause severe burns to skin and eyes. Avoid any skin contact. Effects of contact or inhalation may be delayed. Fire may produce irritating, corrosive and/or toxic gases. Runoff from fire control or dilution water may be corrosive and/or toxic and cause pollution.
| | Fire Hazard Non-combustible, substance itself does not burn but may decompose upon heating to produce corrosive and/or toxic fumes. Some are oxidizers and may ignite combustibles (wood, paper, oil, clothing, etc.). Contact with metals may evolve flammable hydrogen gas. Containers may explode when heated. |
H&Z Industry Co.,Ltd
P.R.China
| solution | 30% in methanol or other types.
| | Appearance | Colorless and transparent viscous liquid
| | Content (total alkali) | 29.5%-31.0%
| | Sodium methoxide(NaOCH3) | 28.9-31.0%
| | Sodium hydroxide(NaOH) | ≤ 0.5%
| | Sodium carbonate(Na2CO3) | ≤ 0.1%
| | Moisture(H2O) | ≤ 0.2%
| | Methanol | 69.0-70.7% |
Xingrui Industry Co., Limited
P.R.China
Hangzhou Meite Industry Co., Ltd (Hangzhou Meite Chemical Co., Ltd)
P.R.China
Leap Chem Co., Ltd
P.R.China
Molecular Formula: CH3NaO
Molecular Weight: 54.02
Hazard Symbols: F,T,C
UN Number: UN 1431 4.2/PG 2
WGKGermany: 1
PackingGroup: II
HS Code: 29051900
Molecular Formula: CH3NaO
BASF SE
Germany
Richman Chemical Inc.,
USA
ICC Chemical Corporation
USA
IBIS Chemie International
India
Wuhan PharmChem Co., LTD.
P.R.China
EINECS:204-699-5
Molecular formula:CH3ONa
Anderson & Steinssen Inc
USA
Shandong Ench Chemical Co.,Ltd
P.R.China
Shandong Lanhai Industry Co.,Ltd.
P.R.China
Hangzhou Zhongqi Chem Co., Ltd
P.R.China
Skyrun Industrial Co., Ltd.
P.R.China
Shandong SanYoung Industry Co., Ltd
P.R.China
Autech Industry Co.,Limited
P.R.China
AK Scientific, Inc.
USA
BuGuCh & Partners
Germany
Evonik Industries AG
Germany
BASF Belgium S.A. / N.V.
Belgium
Merck Schuchardt OHG
Germany
CM Fine Chemicals
Switzerland
|